HEALTH chiefs will defy legal threats to offer elderly patients suffering from a serious eye condition the choice of an alternative, cheaper drug in a move which could save the NHS millions of pounds.
Clinical Commissioning Groups across the North-East have joined forces to agree the use of Avastin for wet age-related macular degeneration (wet AMD), which is a common cause of blindness.
The drug is a tenth of the cost of the alternatives currently in use, Lucentis and Eylea, and could save the region’s financially-stretched NHS up to £13.5m a year – the cost of an extra 270 nurses.
But pharmaceutical companies Novartis, who hold the European rights for Lucentis, and Bayer, who manufacture Eylea, have threatened a judicial review in a bid to force their continued use, claiming the policy breaches patients’ legal right to approved drugs.
Unlike with Lucentis and Eylea, Avastin, which is also used as a cancer drug, is not specifically licensed in the UK for wet AMD treatment, but large-scale international clinical trials have shown that it is just as safe and effective.
Dr David Hambleton, chief officer with the South Tyneside CCG and clinical lead on the matter on behalf of the North East and North Cumbria CCG Forum, said pharmaceutical firms should not hold the NHS to ransom over the issue by dictating which drugs could be used.
He said: “There is a mountain of evidence internationally showing that Avastin is safe and clinically effective for wet AMD.
“We want patients to have the chance to discuss their options with their doctor and make their own choice.
“If a patient chooses to be treated with Eylea or Lucentis instead, the NHS will respect that choice.
“Clinical safety and effectiveness are paramount and we also have a duty to use NHS resources efficiently.”
Dr Hambleton said specialist legal advice had been taken and he was confident the CCGs were acting lawfully, pointing out that Avastin was already being offered to private eye patients and is routinely in use around the world to treat wet AMD.
He also said it was common practice for doctors to prescribe so-called ‘off label’ drugs not for their licensed use and misleading to suggest this was unsafe.
Dr Hambleton added: “Our legal position is strongly supported by a preliminary decision of the European Court handed down in September.
“That is why we have responded robustly to Bayer and Novartis who have threatened a judicial review in an attempt to deny NHS patients this choice.
“It is right to offer our patients this choice, because the beneficiaries will be their friends, neighbours and family members across our region.
“We have no interest in legal disputes, but we cannot allow the NHS to be held to ransom.”
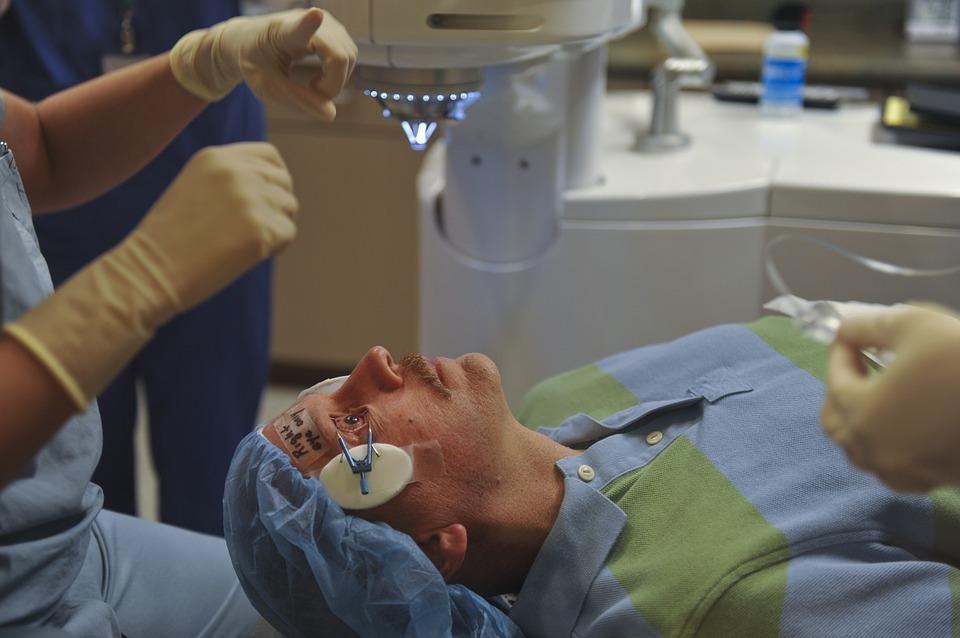
About two thousand people each year in the North-East are newly diagnosed with wet AMD, which causes vision to become increasingly blurred.
All three drugs used to treat the condition and restore sight are delivered via a series of injections.
Bayer and Novartis say that using Avastin instead of licensed options undermines the regulatory framework in place.
A spokesman for Bayer said: “Bayer feels it has to act to challenge the decision taken by these CCGS.”





Comments: Our rules
We want our comments to be a lively and valuable part of our community - a place where readers can debate and engage with the most important local issues. The ability to comment on our stories is a privilege, not a right, however, and that privilege may be withdrawn if it is abused or misused.
Please report any comments that break our rules.
Read the rules here